- Blog
- Youtube music to mp3 download free
- Virtualbox windows 10 64 bit download
- Periodic table valence electron configuration
- Family christmas movies on netflix
- Lifeboat network anti hack
- Test my internet connection stability
- Corolla car maintenance checklist by mileage
- Download play store apps to pc
- Xforce keygen autocad 2012 64 bit
- Logitech g29 logitech gaming software
- Download old manycam
- Siberian mouse pussy girl
- Iomega floppy plus windows 7 drivers
- Xforce keygen autocad 2016 64 bits
- Bioshock 2 remaster trainer
- Mangal font word 365
- Idm full version free download crack
- Twilight forest mod 1-8-9 download
- Naruto shippuden episode 153 english dubbed download
- Siberian mouse mm27 2 girls 1 boy
- In keno what numbers hit the most
- Total war rome pc download torrent
- Scribblenauts remix apk data 5-80
- John deere mini excavator buckets for sale
- Best free business expense tracker app
- Gantt project planner template for excel
- Retroarch gamegear border
- Ps3 move eye camera pc driver
- Checksoft designer software 2007 free download
- Manycam download old version
- Logitech setpoint mouse and keyboard software download
- Ufc 3 pc license key free download
- Sony vegas pro serial number
- Family cashflow
- Microsoft word newspaper template free download
- Iphone unlock software torrent
- Hentai video websites
- Kindergarten pe games
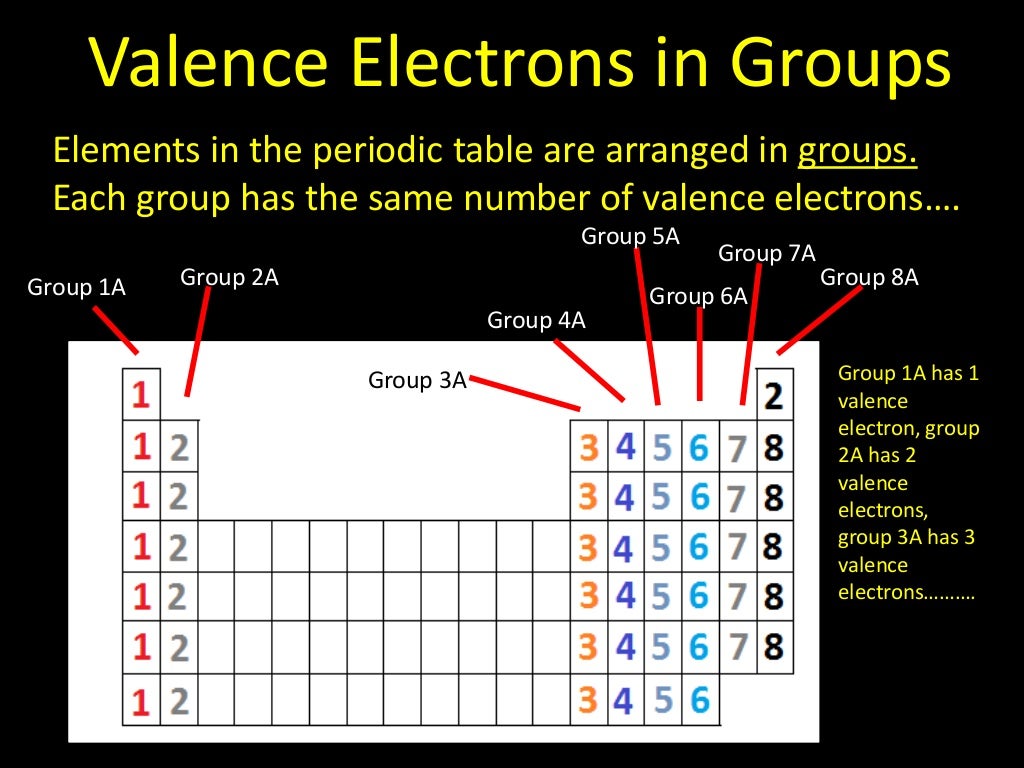
We know that as we scan down a group, the principal quantum number, n, increases by one for each element. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. General trends noted are increasing circle size moving from top to bottom in a group, with a general tendency toward increasing atomic radii toward the lower left corner of the periodic table. 5.2: Electron Configurations, Valence Electrons, and the Periodic Table.

Follow the Aufbau principle and fill electron shells: 1s 2 2s 2 2p 6 Write the noble gas configuration using the noble gas core before neon on the periodic table, followed by the valence electrons. No spheres are provided for the noble or inert gas, group 18 elements. First, use the periodic table and see the number of electrons for a neon atom is 10. Beneath the molecule is the label, “I radius equals 266 p m divided by 2 equals 133 p m.” In figure b, a periodic table layout is used to compare relative sizes of atoms using green spheres. The distance between the radii is 266 p m. Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm.” The fourth diatomic molecule is in purple. The distance between the radii is 228 p m. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. The distance between the radii is 198 p m. The second diatomic molecule is in a darker shade of green. Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. Two spheres are pushed very tightly together. The first model, in light green, is used to find the F atom radius. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The general trend is that radii increase down a group and decrease across a period. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. \): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond.
- Blog
- Youtube music to mp3 download free
- Virtualbox windows 10 64 bit download
- Periodic table valence electron configuration
- Family christmas movies on netflix
- Lifeboat network anti hack
- Test my internet connection stability
- Corolla car maintenance checklist by mileage
- Download play store apps to pc
- Xforce keygen autocad 2012 64 bit
- Logitech g29 logitech gaming software
- Download old manycam
- Siberian mouse pussy girl
- Iomega floppy plus windows 7 drivers
- Xforce keygen autocad 2016 64 bits
- Bioshock 2 remaster trainer
- Mangal font word 365
- Idm full version free download crack
- Twilight forest mod 1-8-9 download
- Naruto shippuden episode 153 english dubbed download
- Siberian mouse mm27 2 girls 1 boy
- In keno what numbers hit the most
- Total war rome pc download torrent
- Scribblenauts remix apk data 5-80
- John deere mini excavator buckets for sale
- Best free business expense tracker app
- Gantt project planner template for excel
- Retroarch gamegear border
- Ps3 move eye camera pc driver
- Checksoft designer software 2007 free download
- Manycam download old version
- Logitech setpoint mouse and keyboard software download
- Ufc 3 pc license key free download
- Sony vegas pro serial number
- Family cashflow
- Microsoft word newspaper template free download
- Iphone unlock software torrent
- Hentai video websites
- Kindergarten pe games
